
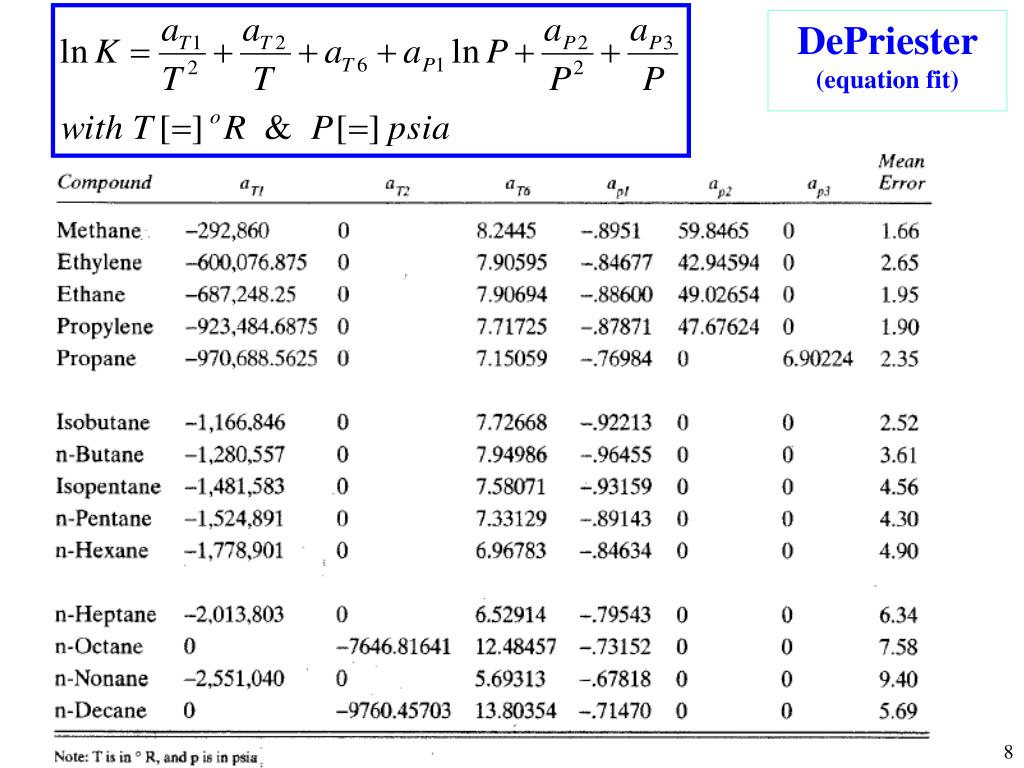
Boiling point - the temperature at which a liquid turns into a gasįor hydrocarbons with the same carbon number the boiling point increases in the following order:.Melting point - the temperature at which a solid turns into a liquid.More detailed definitions and examples of molecular structures of the different groups are given below the figures. Campbell and Company, Norman, Oklahoma, USA, 2001.The tables and figures below show how the boiling point changes with increasing carbon number up to C 33 for different kinds of hydrocarbons, alcohols and carboxylic acids. M., “Gas conditioning and processing, Volume 2: Equipment Modules,” John M. Campbell and Company, Norman, Oklahoma, USA, 2001. “Gas conditioning and processing, Volume 1: Fundamentals,” John M. Campbell and Company, Norman, Oklahoma, USA, 1994. Lilly, “Gas conditioning and processing, Volume 3: Advanced Techniques and Applications,” John M. To learn more on applications of K-values and their impact on facilities calculation, design and surveillance, refer to JMC books and enroll in our G4 (Gas Conditioning and Processing) and G5 (Gas Conditioning and Processing – Special) courses. Many less than desirable systems results from failure to recognize this.
#Depriester chart low temperature series#
This is important, for in many systems a series of VLE calculations is made the output from one is the input to another. The compositional analysis on which the calculation was based will often be in error more than this. There are several methods available for this purpose (See pages 113-116 of Vol 1 of JMC Books).Īn experienced practitioner usually can predict the quantity of a specified liquid within 6% (for a specified analysis and conditions). It is most important that the K-values be internally consistent.
It is doubtful if one ever will encounter the analyses, flow rates and exact other conditions used as the design basis. This range, rather than one set of “magic” numbers, is then used to size equipment. Generally, crude oil and NGL phase behavior is handled by different models.Īll K-values are sensitive to composition, particularly the very volatile components like nitrogen, methane, and ethane.įor design purposes, several models may be used to determine a range of results. An experienced practitioner may have two or three different models or program available. There is no single K-Value correlation that is superior for all mixtures encountered. The accuracy of the results of calculations involving K-Values depends on the reliability of sampling, of the analysis of that sample, and the K-Value correlation used. Therefore, the following guidelines extracted from page 128 of Vol 1 of JMC book are suggested. Again, the calculated liquid fractions by the Raoult’s law and Wilson correlations are close to each other but they deviate considerably from the GPA charts and the SRK EoS results.ĭue to the observation made in the previous section and other studies, care must be taken in selecting K-values correlations. Similarly, for the same mixture shown in Table 1, a series of flash calculations for two isotherms were performed and the calculated liquid fractions (L/F) using different methods are compared in Table 5.
